The importance of placental research
Pregnancy and childbirth remain major causes of female mortality, and over half a million women die each year as a direct consequence. Hence, improving maternal health was designated the 5th of the Millennium Development Goals, with the aim of reducing maternal mortality between 1990 and 2015 by three quarters. Sadly, it is the area of least progress among all the Goals. The majority of these deaths occur in low- and middle-income countries through complications such as obstructed labour, infection and haemorrhage, and should be preventable by improved obstetric care. However, it remains the case that the hypertensive disorders pre-eclampsia and eclampsia, for which there are currently no treatments, account for 10-15% of maternal deaths. These disorders are also associated with increased infant mortality. In the UK, perinatal mortality remains in the region of 5-10 deaths per 1,000 potentially viable infants. Stillbirth affects 1 in 200 births after 24 weeks (approximately 4,000 per annum), and the majority are related to placental dysfunction. Elective preterm delivery is also frequently related to abnormal placentation, and accounts for approximately 60% of babies weighing less than 1000g. This group has a 40-45% rate of severe neurodevelopmental impairment. Hence, the consequences of defective placentation include mortality and severe morbidity, and associated health care costs both in the short and long term.
There is thus an urgent and persuasive need for more research into the development and function of the placenta.
Our scientific approach
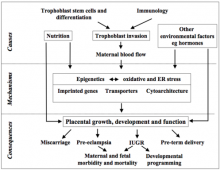
Placental biology spans a broad range of disciplines, but all involve an understanding of the trophoblast cell lineage, a unique mammalian cell type that interfaces with the maternal tissues. As the name indicates (trophos Gr. nourishment), trophoblast evolved with the principal role of supplying nutrients to support growth of the fetus. Trophoblast is the first cell lineage to differentiate after fertilization, and its sub-populations play key roles in placental development and function. In the human this is complicated by the fact that the trophoblast is highly invasive, eroding into the maternal arteries in order to secure a blood supply to the placenta. This process poses immunological challenges, as the genome of the placental cells contains both maternal and paternal contributions. Many common complications of pregnancy are rooted in defects in trophoblast invasion during early pregnancy, and the ensuing compromise in maternal blood flow to the placenta. The organ fulfills a remarkable variety of vital roles, including the transport of oxygen and nutrients, acting as an endocrine organ that influences maternal physiology and behaviour, forming a shield against attack by the mother’s immune system, protecting the fetus from maternal sex and stress hormones, and constituting a barrier against the vertical transmission of pathogens. Disruption of any one of these functions may impact adversely on fetal development, and consequently we take a uniquely integrated and cross-disciplinary approach.

